 The Florida Hospital Orthopaedic Institute’s Research Department is looking for people to take part in a trial for innovative, nonsurgical, investigational treatment for knee pain and function loss caused by osteoarthritis (OA).
The Florida Hospital Orthopaedic Institute’s Research Department is looking for people to take part in a trial for innovative, nonsurgical, investigational treatment for knee pain and function loss caused by osteoarthritis (OA).
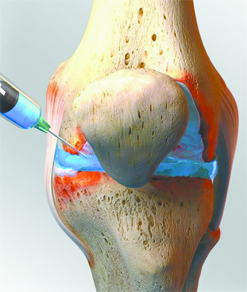
Osteoarthritis is a common type of arthritis that can often affect the knee. It is a complex disease involving wear and tear degeneration and inflammation and can be a major source of disability due to pain and loss of function. If you have knee osteoarthritis, there are a variety of treatments to relieve your symptoms temporarily. Conservative treatment can include NSAIDS, weight reduction, hot/cold therapy, and physical therapy. Injections are usually reserved until conservative measures have not provided satisfactory relief, and are offered as one of the last non-operative treatments. The two most commonly used knee injections are hyaluronic acid and corticosteroids, but other options are available.
Corticosteroid
Corticosteroids have an anti-inflammatory effect and are often used for managing flare-ups of OA pain and swelling with fluid buildup in the knee. (1) Corticosteroid injections help to temporarily relieve symptoms by decreasing pain, swelling and inflammation in the joint. They work quickly, but the relief often lasts from only a few days to perhaps six months. However, repeated steroid injections into the joint may actually contribute to cartilage breakdown, and patients will likely be limited in the number of injections received in the same joint. (2) The American College of Rheumatology subcommittee on OA recommends corticosteroid injections as an effective method of decreasing pain. (3)
Hyaluronic Acid
Hyaluronic Acid is what makes up most of the fluid in the knee joint and can break down when you have OA in the joint. It acts as a shock absorber and lubricant, allowing joints to move smoothly over each other. Hyaluronic acid is typically produced from harvested rooster combs, and can be injected into the joint to lessen pain and inflammation. It often requires weekly injections, depending on the product used, for three to five weeks. (2) However, the American Society of Orthopedic Surgeons does not recommend using Hyaluronic Acid injections for patients with symptomatic OA of the knee. (4)
Platelet-Rich Plasma (PRP)
Although experimental, Platelet-Rich Plasma (PRP) injections have been used for patients with knee osteoarthritis. PRP is a platelet concentration from normal blood, and some compositions are also filtered to remove white blood cells. Not all studies support the use of PRP to treat osteoarthritis, and FDA has not approved any PRP for this indication. Research has shown that knees treated with 1 or 2 PRP injections saw a reduction in pain and stiffness, as well as improvement in knee function, but after 6 months, positive results declined. (5) Research data does not indicate that PRP treatment will cause cartilage and meniscus to regenerate in patients with substantial cartilage damage, but promising results of symptomatic improvement have been shown, particularly in younger patients with mild OA cases. (6)
Autologous Protein Solution (APS)
Another investigative method of treatment is being studied by Florida Hospital’s Orthopaedic Research Department in hopes of offering an alternative knee injection that uses an autologous protein solution (APS), which contains growth factors and proteins found in the patient’s own blood. (7) The use of APS to treat cells involved in the degenerative process of OA is innovative and represents a more advanced approach than other blood-based treatments, such as PRP. In comparison to other types of injections to treat knee OA, the APS concentration being investigated is thought to specifically target the reduction of inflammation and may promote tissue repair within the joint, according to in vitro and animal studies. (8,9)
PROGRESS IV clinical trial – nSTRIDE® APS Kit
To investigate this treatment, the research team is conducting a double-blind study called the PROGRESS IV clinical trial, sponsored by Zimmer Biomet, a global medical device company. The PROGRESS IV clinical trial is testing a device called the nSTRIDE® APS Kit. The trial will focus on people with osteoarthritis symptoms in one knee only and will study the potential to alleviate pain and restore loss of knee function associated with OA. Additionally, X-ray and MRI is being used to see if the APS could possibly help the tissue inside the joint repair itself.
The study has inclusion and exclusion criteria that must be met in order to qualify for participation. Conditions, other than OA, that affect the ability to ambulate or other untreated joint injuries may exclude patients from the study. Eligible patients are enrolled and randomly selected with a 50/50 chance to receive the APS treatment or saline, and patients will not know which injection they received.
For anyone who does not meet the criteria for the study, Dr. Cole’s practice will work with them to find alternative treatment options for managing their osteoarthritis. People may or may not benefit from taking part in the trial, but information learned in the study may help patients with osteoarthritis in the future. All of the study procedures and risks associated with the study will be discussed with potential candidates before a decision to participate is made.
How can I get more information about this study?
For further information regarding the study, or to determine if you meet eligibility criteria, text 1KNEE to the number 87888, call (773) 313-3077, or visit www.zimmerbiomet.com/nstridetrial
Dr. J. Dean Cole specializes in orthopaedic traumatology and joint replacements at the Florida Hospital Orthopaedic Institute Fracture Care Center.
For more information, go to www.FractureCareCenter.com or call 407-895- 8890.
1. “Injectable Medications for Knee Osteoarthritis.” WebMD, www.webmd.com/osteoarthritis/features/injections-for=osteoarthritis-pain. Accessed 05 December 2017.
2. “Injections and Procedures for Knee Pain.” Arthritis Foundation, http://www.arthritis.org/about-arthritis/where-it-hurts-knee-pain/treatment/knee-injection.php. Accessed 05 December 2017.
3. Hochbert MC, et al. American College of Rheumatology 2012 recommendations for the use of nonpharmacologic and pharmacologic therapies in osteoarthritis of the hand, hip, and knee. Arthritis Care Res (Hoboken) 2012; 65:465-474.
4. Jevsevar DS, et al. The American Academy of Orthopaedic Surgeons evidence-based guideline on: treatment of osteoarthritis of the knee, 2nd edition. J Bone Joint Surg Am. 2013; 95: 1885-1886.
5. “Efficacy of Platelet-Rich Plasma Injections.” Arthritis-health, https://www.arthritis-health.com/treatment/injections/efficacy-platelet-rich-plasma-injections. Accessed 05 December 2017.
6. Ayhan E, Kesmezacar H, Akgun I. Intraarticular injections (corticosteroid, hyaluronic acid, platelet rich plasma) for the knee osteoarthritis. World J Orthop. 2014; 5(3):351-361.
7. Matuska A, et al. Autologous solution protects bovine cartilage explants from IL-1α- and TNFα-induced cartilage degradation. J Orthop Res. 2013;31(12): 1929-1935.
8. O’Shaughnessey KM, et al. Blood derived anti-inflammatory protein solution blocks the effect of IL-1β on human macrophages in vitro. Inflamm. Res. 2011;60: 929.
9. King W, et al. Human blood-based anti-inflammatory solution inhibits osteoarthritis progression in a meniscal-tear rat study. J Orthop Res. (2017). DOI 10.1002/jor.23528.
Check Also
Obstructive Sleep Apnea & Oral Appliances: A Solution for a Good Night’s Sleep
By Richard W. Rozensky, DDS, D.ABDSM Sleep apnea affects more than 25 million people in …
 Central Florida Health and Wellness Magazine Health and Wellness Articles of the Villages
Central Florida Health and Wellness Magazine Health and Wellness Articles of the Villages